In May, NMPF told the U.S. Food and Drug Administration (FDA) that the agency needs to adopt a prudent approach on dairy equivalence issues that will be supportive of U.S. dairy exports. At issue is FDA’s determination of whether a foreign country has “equivalent” food safety parameters to the United States, such as those followed by dairy farmers and processors to ensure consumers receive safe milk products.
Earlier this spring, in its first-ever equivalence action, FDA determined that European Union (EU) shellfish are as safe as those harvested in the United States, and as a result recommended granting equivalence to certain types of raw shellfish coming from the EU. This determination is meant to restart trade after an eight-year stalemate that began when the EU abruptly cut off access to its market for U.S. shellfish.
NMPF expressed concerns that the manner in which the U.S. government is handling this shellfish trade topic would pose a deep concern if that same process were applied to the trade of dairy foods. National Milk raised the issue in light of the publication this spring of FDA’s shellfish equivalence determination, and ongoing “Grade A” dairy equivalence assessments of New Zealand, Canada and various countries in the European Union (EU).
In detailed comments to FDA, NMPF and the U.S. Dairy Export Council (USDEC) pointed out that it would be extremely problematic if the agency applied the same process to the dairy industry, as that approach would run counter to nationally harmonized food safety regulations for dairy, fall short of upholding high U.S. consumer protections, and fail to sufficiently tackle barriers to U.S. dairy exports, among other concerns.
NMPF also met with Dr. Stephen Ostroff, deputy commissioner of FDA, and numerous FDA staff working on dairy and trade issues to explain why the U.S. dairy industry has concerns, and to pledge to work with FDA to address these worries by pursuing a more workable approach to tackling dairy trade issues, including those pertaining to equivalence. In a follow-up, NMPF and USDEC jointly sent a letter to FDA Commissioner Scott Gottlieb to reiterate dairy’s concerns and commitment to collaborating with FDA and its interagency partners.

 The U.S. Department of Agriculture (USDA) announced on June 7 that California dairy producers approved a Federal Milk Marketing Order (FMMO) for the entire state of California.
The U.S. Department of Agriculture (USDA) announced on June 7 that California dairy producers approved a Federal Milk Marketing Order (FMMO) for the entire state of California.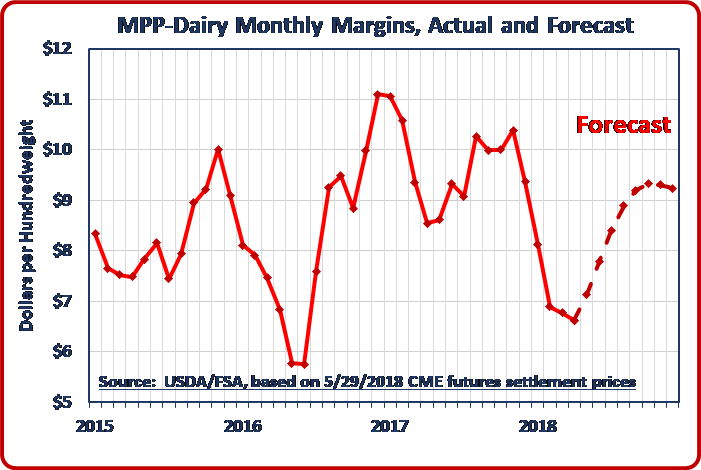 Under the dairy Margin Protection Program (MPP), the monthly margin for April 2018 decreased an additional $0.15 per hundredweight from March, to $6.62/cwt. This was the fifth consecutive drop in the MPP monthly margin. The April all-milk price increased by $0.20/cwt. from March, to $15.80/cwt. The April feed cost formula was up by $0.35/cwt. from March, with most of the increase – on a per-hundredweight-of-milk basis – due to an increase in the cost of alfalfa hay. The prices of corn and soybean meal also contributed smaller increases to the MPP feed cost formula calculation.
Under the dairy Margin Protection Program (MPP), the monthly margin for April 2018 decreased an additional $0.15 per hundredweight from March, to $6.62/cwt. This was the fifth consecutive drop in the MPP monthly margin. The April all-milk price increased by $0.20/cwt. from March, to $15.80/cwt. The April feed cost formula was up by $0.35/cwt. from March, with most of the increase – on a per-hundredweight-of-milk basis – due to an increase in the cost of alfalfa hay. The prices of corn and soybean meal also contributed smaller increases to the MPP feed cost formula calculation. An amendment to the 2018 House Farm Bill that would have allowed the interstate sale of unpasteurized milk was soundly defeated thanks to strong opposition from National Milk, its member cooperatives, several other key industry stakeholders, as well as consumer and public health advocates.
An amendment to the 2018 House Farm Bill that would have allowed the interstate sale of unpasteurized milk was soundly defeated thanks to strong opposition from National Milk, its member cooperatives, several other key industry stakeholders, as well as consumer and public health advocates. NMPF continues to work with its congressional allies to ensure the enactment of a bipartisan, bicameral Farm Bill before the current one expires on Sept. 30. Both the House and Senate are making progress on their respective versions of the Farm Bill, with action likely in both chambers in June.
NMPF continues to work with its congressional allies to ensure the enactment of a bipartisan, bicameral Farm Bill before the current one expires on Sept. 30. Both the House and Senate are making progress on their respective versions of the Farm Bill, with action likely in both chambers in June. The NMPF Board of Directors voted in early June to continue the Cooperatives Working Together (CWT) program, the historic dairy farmer self-help export assistance effort.
The NMPF Board of Directors voted in early June to continue the Cooperatives Working Together (CWT) program, the historic dairy farmer self-help export assistance effort. ARLINGTON, VA – The board of directors of the National Milk Producers Federation (NMPF) today voted to extend funding through 2021 for Cooperatives Working Together (CWT), the farmer-funded export assistance program that assists member cooperatives in exporting dairy products.
ARLINGTON, VA – The board of directors of the National Milk Producers Federation (NMPF) today voted to extend funding through 2021 for Cooperatives Working Together (CWT), the farmer-funded export assistance program that assists member cooperatives in exporting dairy products.



